22-25 Апреля 2020 года в Ханой Вьетнам пройдет строительная конференция. Мы направили туда статью, касательно очистки сточных вод полигонов ТБО. Тема очень важная во всем мире, поэтому Вьетнам с удовольствием принял статью в печать.
Treatment of landfill leachate using reverse osmosis membranes
Tatiana Shirkova, Alexei Pervov and Maria Kiryushina
Moscow State University of Civil Engineering (National Research University), Russia, Moscow, Yaroslavskoye Shosse, 26
E-mail: mabell@bk.ru
Abstract. Two different approaches to treat municipal landfill leachate with reverse osmosis membranes are evaluated and discussed. Investigations are aimed at production of quality water that could be used for industrial use or meet regulations for surface water sources discharge. Also possibilities are evaluated to radically reduce concentrate flow by a value that does not exceed 0,5-1 per cent of initial feed water flow to withdraw all rejected impurities together with dewatered sludge. Experimental research is conducted to develop membrane operational modes during leachate treatment. Experimental procedure is developed and described to evaluate reduction of membrane flux and rejection during leachate treatment and recovery increase. Results of experimental investigations are presented that enable us to determine main characteristics of membrane process such as: membrane flux, ammonia rejection, membrane types, number of stages, recovery. Experimental relationships are obtained that enable us to determine the required membrane recovery values that correspond to ammonia concentration in the feed water to meet required regulation values in the product water. The developed tools to evaluate the required membrane area and required amount of membrane modules (required membrane area) are developed and described. Influence of organics defined as COD (chemical oxygen demand) on membrane performance is investigated. Optimum values of the working pressure are evaluated as well as other economic parameters are determined. Main tools to handle concentrate flow of membrane unit, to withdraw it with dewatered sludge as well as methods to utilize sludge and concentrate are also discussed. To reach the required ammonia concentration value in product water, a double-stage treatment with low pressure reverse osmosis membrane is required.
Keywords: landfill leachate, reverse osmosis, nanofiltration, membrane fouling, recovery.
- Introduction
Construction of landfills is a conventional solution for municipal solid wastes disposal and storage [1]. Generation of leachate by municipal solid waste landfills causes a significant threat to surface and ground water. The landfill leachate contains many organic and inorganic contaminants generated within landfill body [1]. Different methods are applied to remove these contaminants, such as chemical and biological techniques [2]. To remove dissolved salts and biogenic elements (like ammonia and phosphate ions) reverse osmosis (RO) membranes are applied [3]. Often leachate that has high TOC value (from 1000 to 5000 ppm) is subjected to chemical treatment to remove suspended matter and dissolved organics [4]. High organic content increases osmotic pressure and decreases membrane permeability [5]. From the other point, chemical treatment, coagulation, flocculation and further sedimentation require large volumes of water treatment structures and footprint. The goal of the present report was to provide experimental results to develop efficient membrane techniques and to evaluate required membrane area and operational costs. Figure 1 shows a flow diagram of landfill leachate handling and treatment.
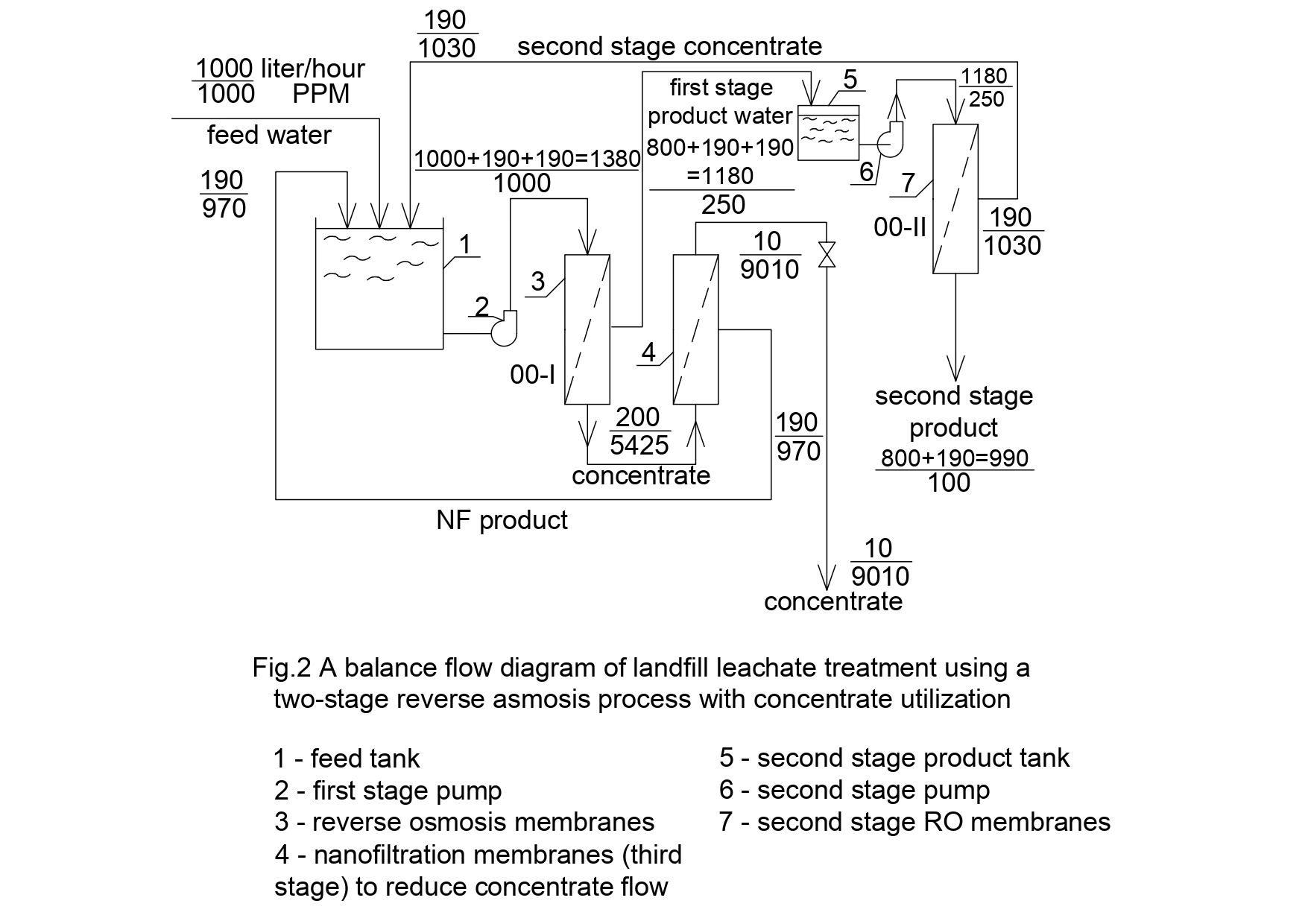
Reverse osmosis is widely applied in many wastewater treatment projects [4,5] due to membrane abilities to reject inorganic (ammonia, phosphates, chlorides) and organic (humid acid, halogenocarbons, phenols) contaminants [5,6]. Often a double-stage membrane treatment technique is required to reach low concentration values of nitrates and ammonia in the product water (Figure 2) to meet strict water discharge regulations [ 2,5]. When landfill leachate is purified using membrane techniques, the problem of concentrate handling and utilization often arise [6].Concentrate flow usually constitutes from 15 to 30 per cent of leachate volume [5,7]. In landfill leachate treatment practice concentrate is returned back to landfill body [2] that can cause increase of different contaminants concentration values with time.
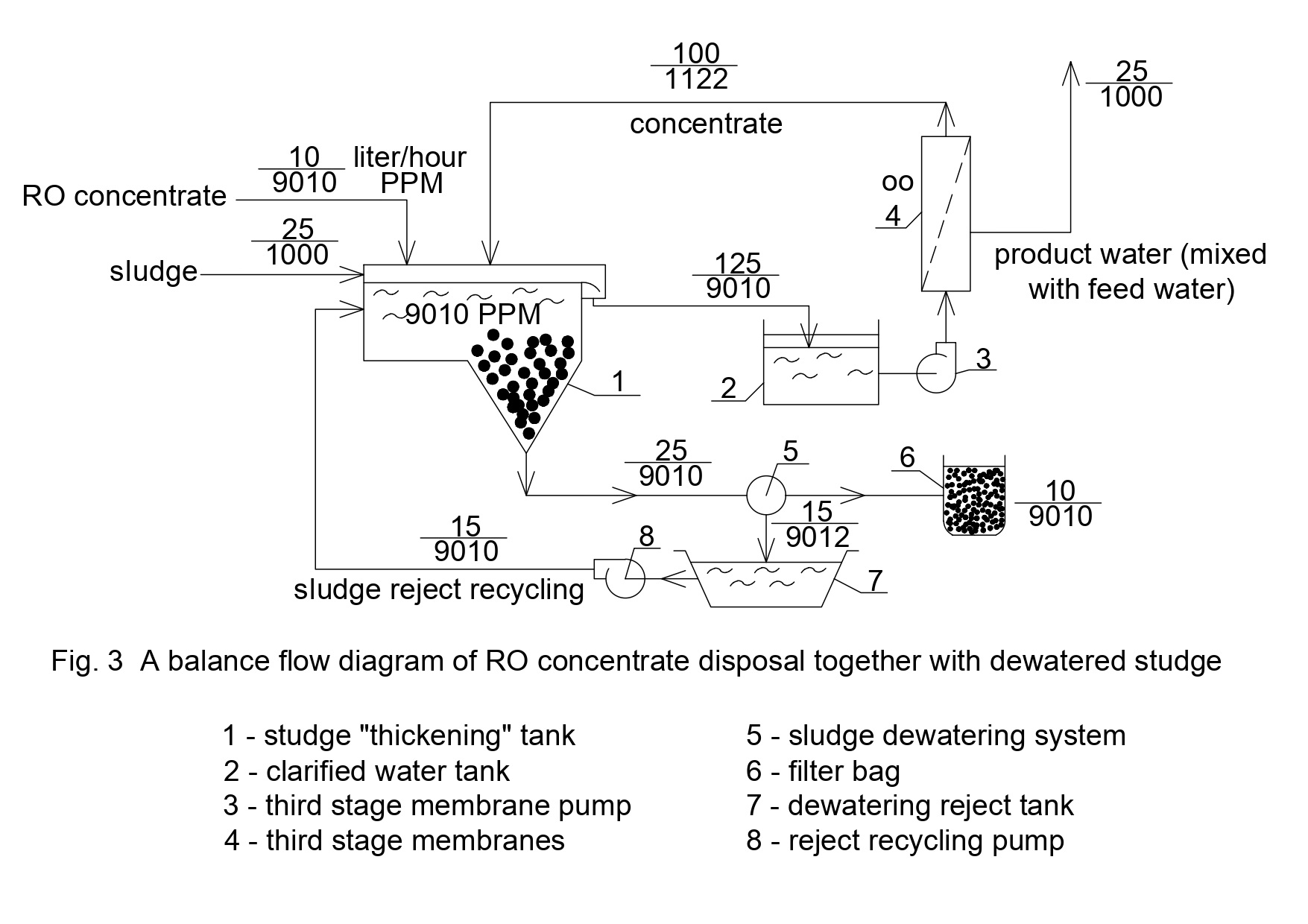
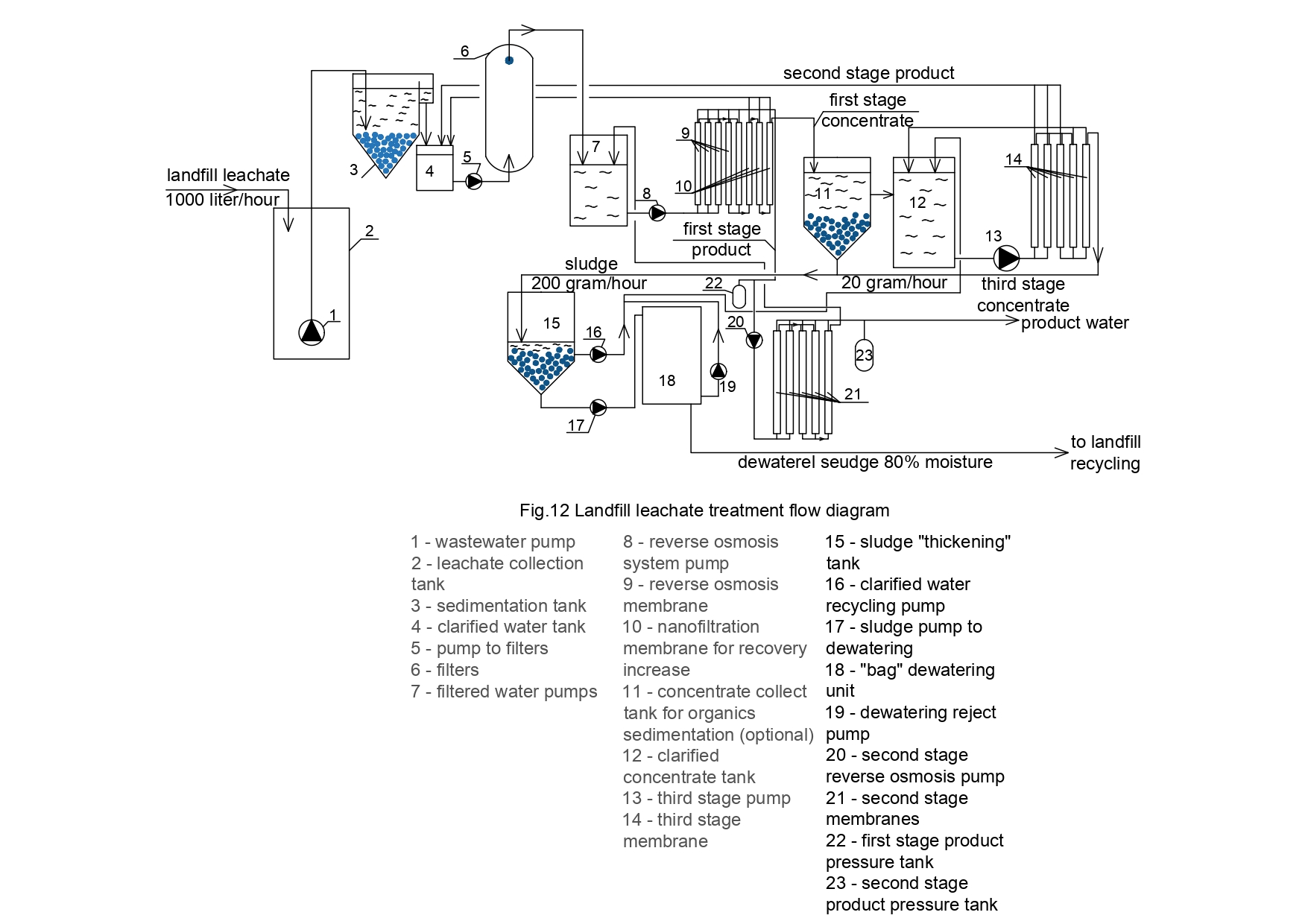
In [9] authors have presented a new technique to reduce concentrate flow to reach 0,3-1,0 percent of initial feed water flow that enters membrane facility. Thus concentrate can be withdrawn and utilized together with dewatered sludge [8,9].In present report authors aimed at the application of approach described in [9] to treat landfill leachate and to produce quality desalinated water and dewatered sludge that can be utilized using verification, incineration or other techniques. The flow diagram of the process of radical concentrate flow reduction and withdrawal together with dewatered sludge is shown on Figure 3.
To pretreat leachate prior to reverse osmosis desalination chemical treatment techniques were applied [3]. As it was discussed in [5,9,10], the presence of high molecular organics in the feed water decreases membrane flux. Sedimentation of coagulated and flocculated organics provides additional amount of sludge that facilitates concentrate utilization: the sludge moisture value of 80 per cent corresponds to water amount in the sludge five times more than amount of solids. Therefore, high TOC values in leachate provide favorable conditions to use the developed techniques [5,10]. For the cases where leachate is directly treated by membranes, research was aimed maximum recovery values to return minimum water back to landfill body (Figure 1).
Present study demonstrates results of experimental evaluation of characteristics and parameters of reverse osmosis process to purify leachate. As concentration values of ammonia in leachate vary from 80 to 2000 ppm, two stages of reverse osmosis membrane treatment were applied (Figure 2).
- Materials and methods
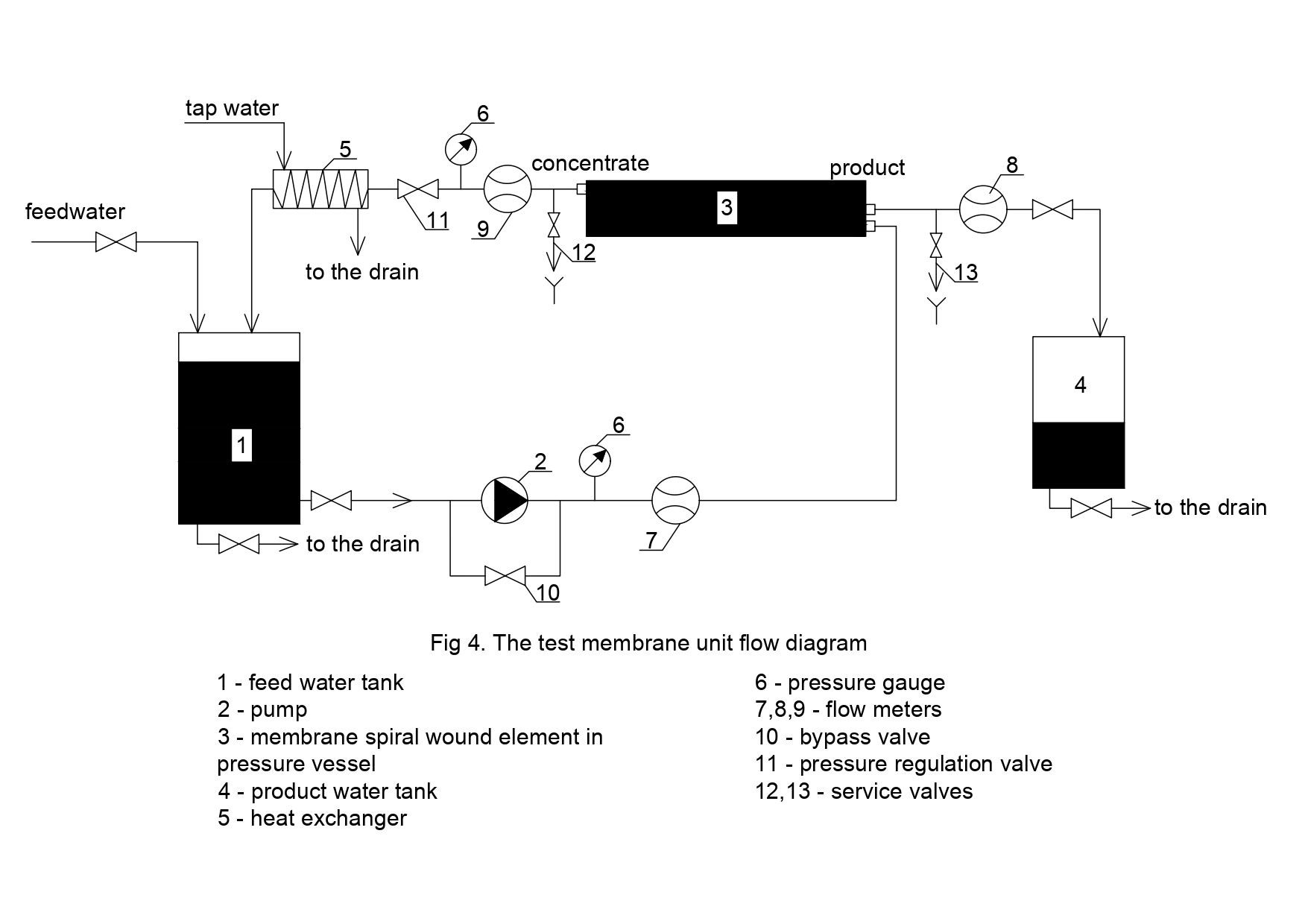
The test unit flow diagram is shown on Figure 4. Wastewater after bioreactor was collected in the feed water tank 1. The pump 2 to the module 3 pumped wastewater. Concentrate was returned back to the tank 1 and product flow was forwarded to product tank 4. Pressure value was regulated using the pressure valve 5 and controlled by pressure gauge 6. Reverse osmosis membrane used were 1812 type elements manufactured by CSM Company, Korea (model 1812 BLN). For concentrate treatment, 90NE membranes were used (model 1812 90NE). The gear pump created pressure. Pressure value was 6 Bars.
Chemical compositions of landfill leachate without chemical treatment, after chemical treatment, after first stage of RO and after second stage of RO are presented in Table 1.
Table 1. Results of chemical analyses of leachate i ∙ treatment.
- Results
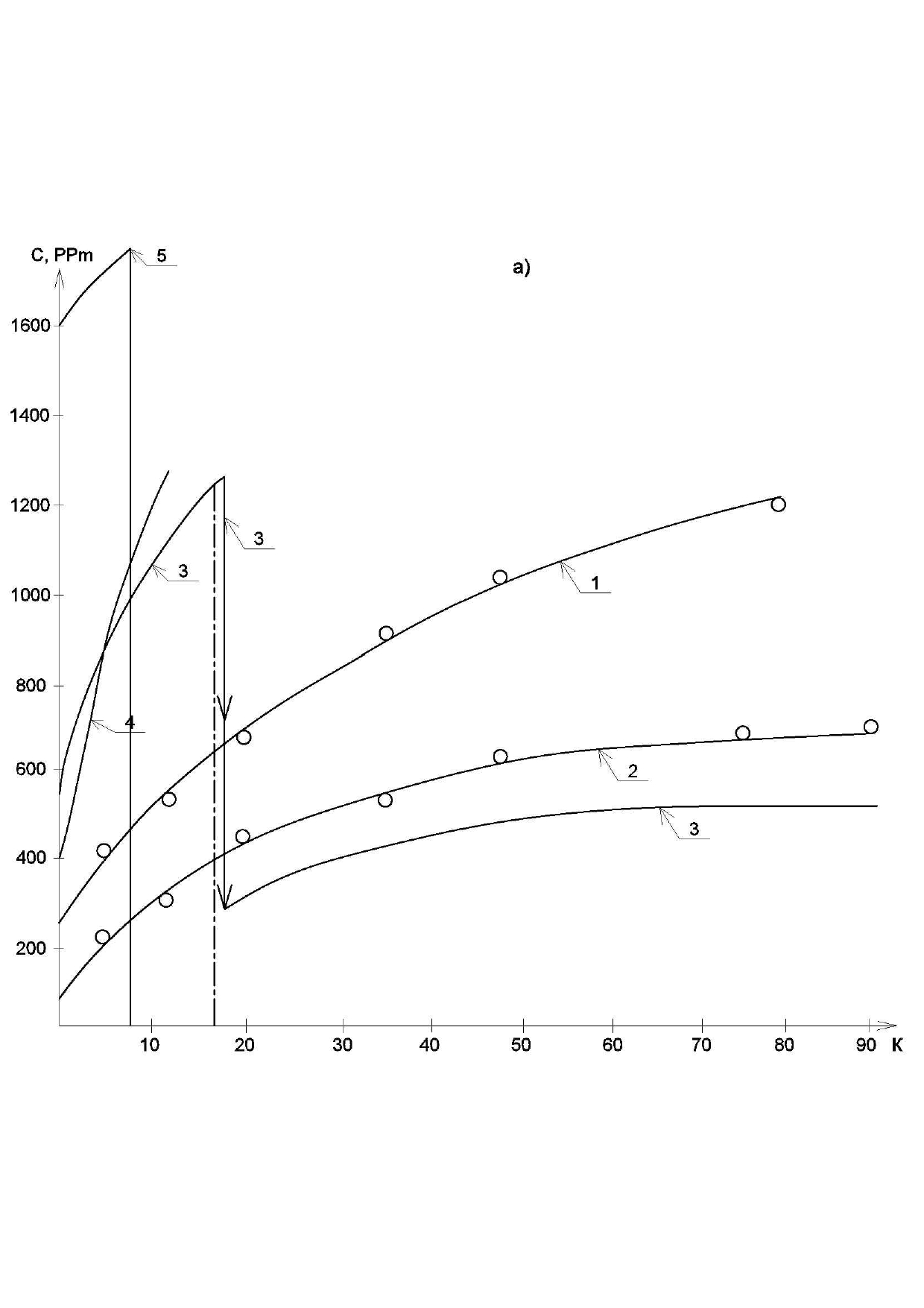
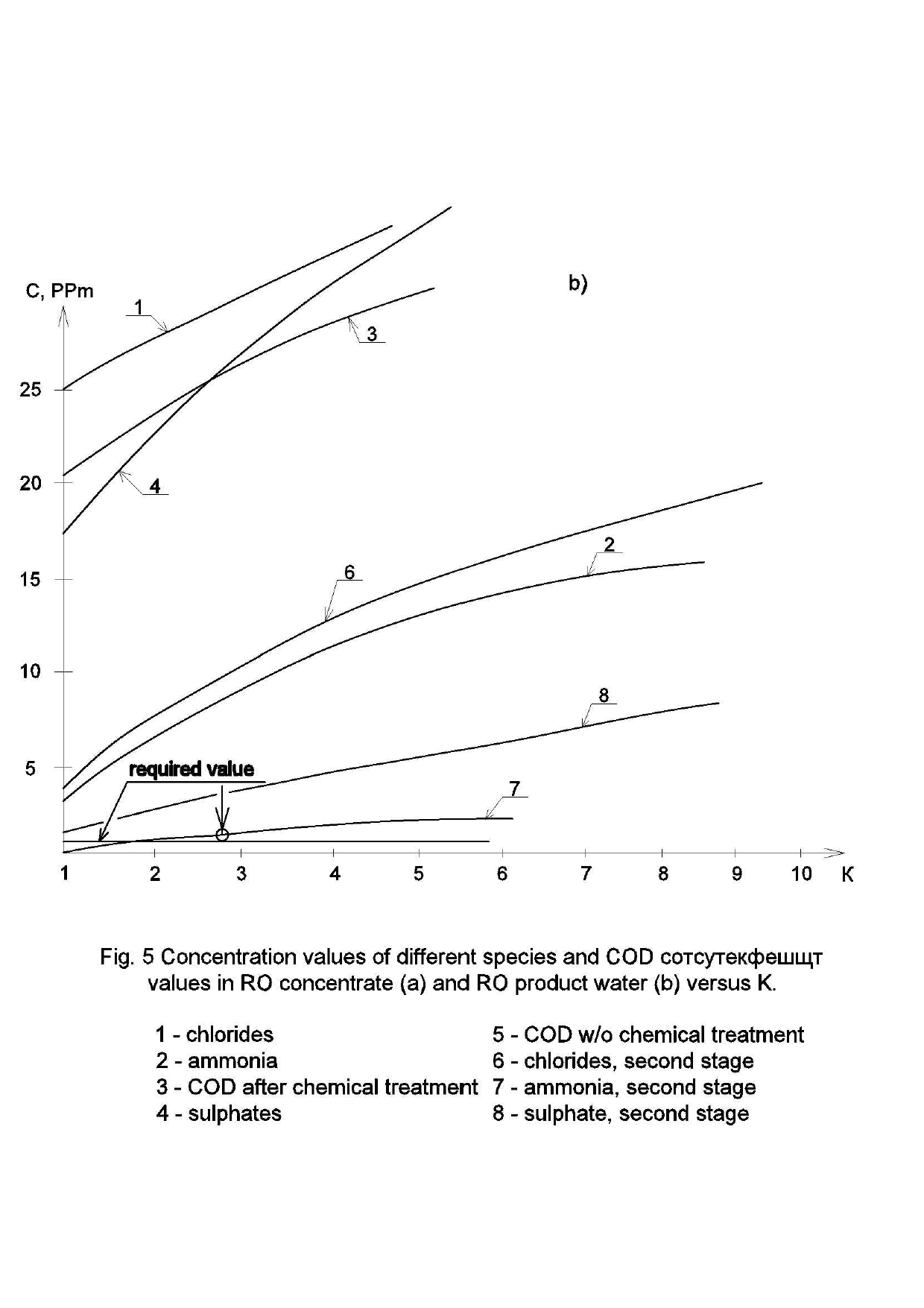
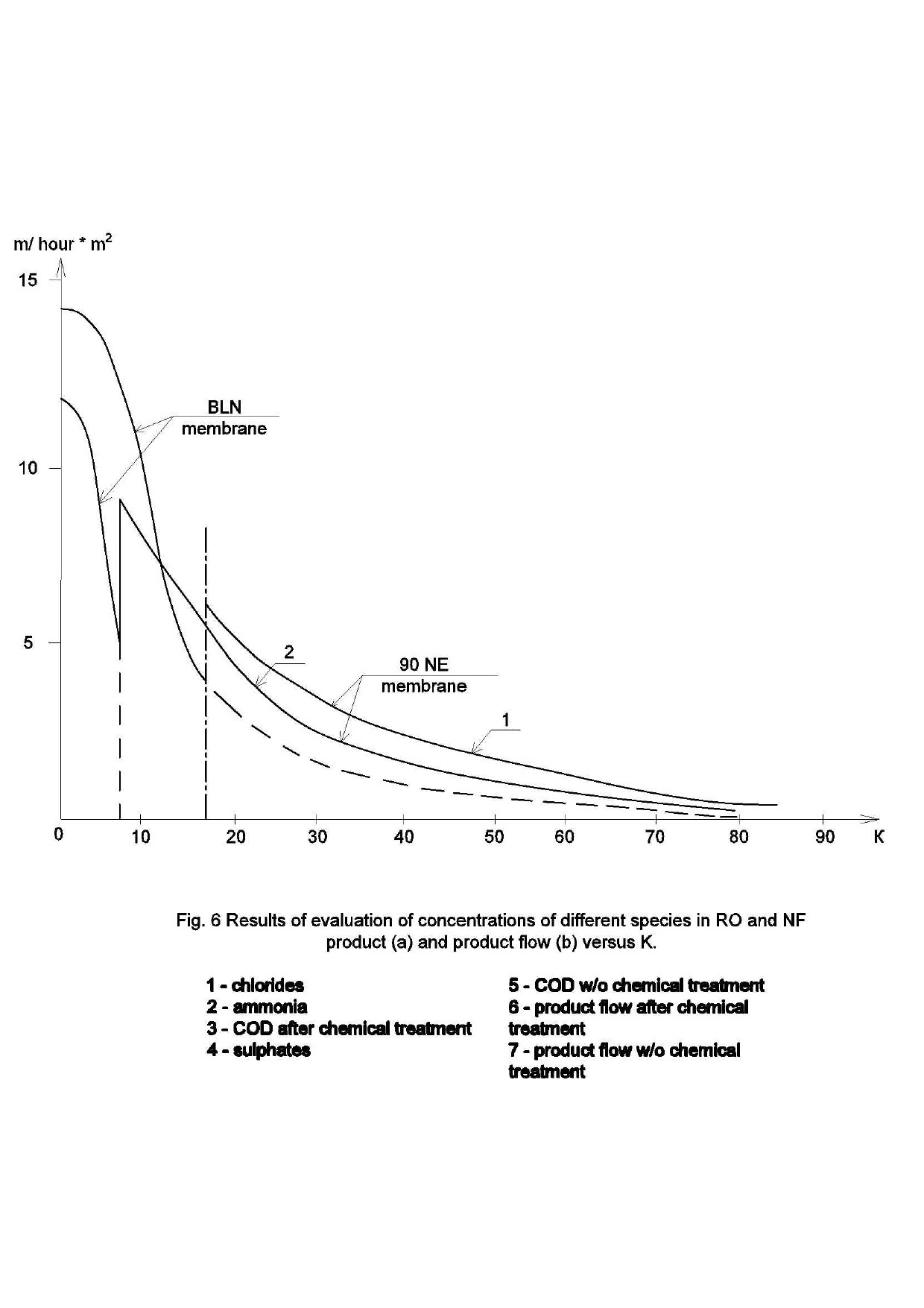
Experimental data was consistently processed with the aim to obtain values of the main technical parameters of membrane unit. First of all, dependencies of various ions concentration values on the recovery and the product volume were determined. Figure 5 shows dependencies of ionic concentrations of sulphates, chlorides and ammonia ions on concentration factor K values , where K is a ratio of the initial volume in tank 1 ( Figure 4) to the volume of product water in product tank (4) during the test run.
At the beginning of experiment, initial volume was 70 liters. This volume was treated on the first stage of experiment by low pressure reverse osmosis BLN type membranes until the amount of water in feed tank 1 ( Figure 4) was 7 liters and the volume of product water collected in tank 4 was 63 liters. On the second stage of experiment the remained amount of concentrate (7 liters) was treated using nanofiltration 90-NE membranes that demonstrated higher product flow.
Then, decrease of membrane product flow throughout the experiment was evaluated as a dependence on K value (Figures 6-8).
Figure 5(a) shows dependencies of ammonia and sulphate ionic concentrations as well as COD (chemical oxygen demand) values in concentrate flow during experimental test run. At different stages (that corresponded to certain K values, such as: 2, 4, 6, 10, 20, 40, 80)product water samples were withdrawn where concentrations of all impurities as well as TDS values were determined.
Similar dependencies of different species’ concentration values in product water and concentrate versus K are shown on Figure 5. Figure 5(a) shows increase of different rejected by membrane impurities versus K.
As it is shown on Figure 5(b), ammonia concentration values in product are higher, than discharge regulation values. This can be explained by the low rejection of monovalent ions by low-pressure RO membranes in the pressure range of 6-12 Bars. Ammonia ion rejection in this pressure range varies between 85 and 90 per cent. Therefore, to arrange high rejection of ammonia, the second stage RO BLN membrane treatment should be applied.
Figure 6 shows decrease of membrane product flow versus K on both stages of concentrate treatment. Product flow was measured under pressure value of 6 Bars.
- Discussions and conclusions
The next step was devoted to determination of technical parameters of membrane unit.
Experimental data processing was aimed at determination of technical characteristics of membrane unit, such as membrane area required and minimal concentrate amount.
To predict influence of membrane recovery on product water quality, various relationships were examined.
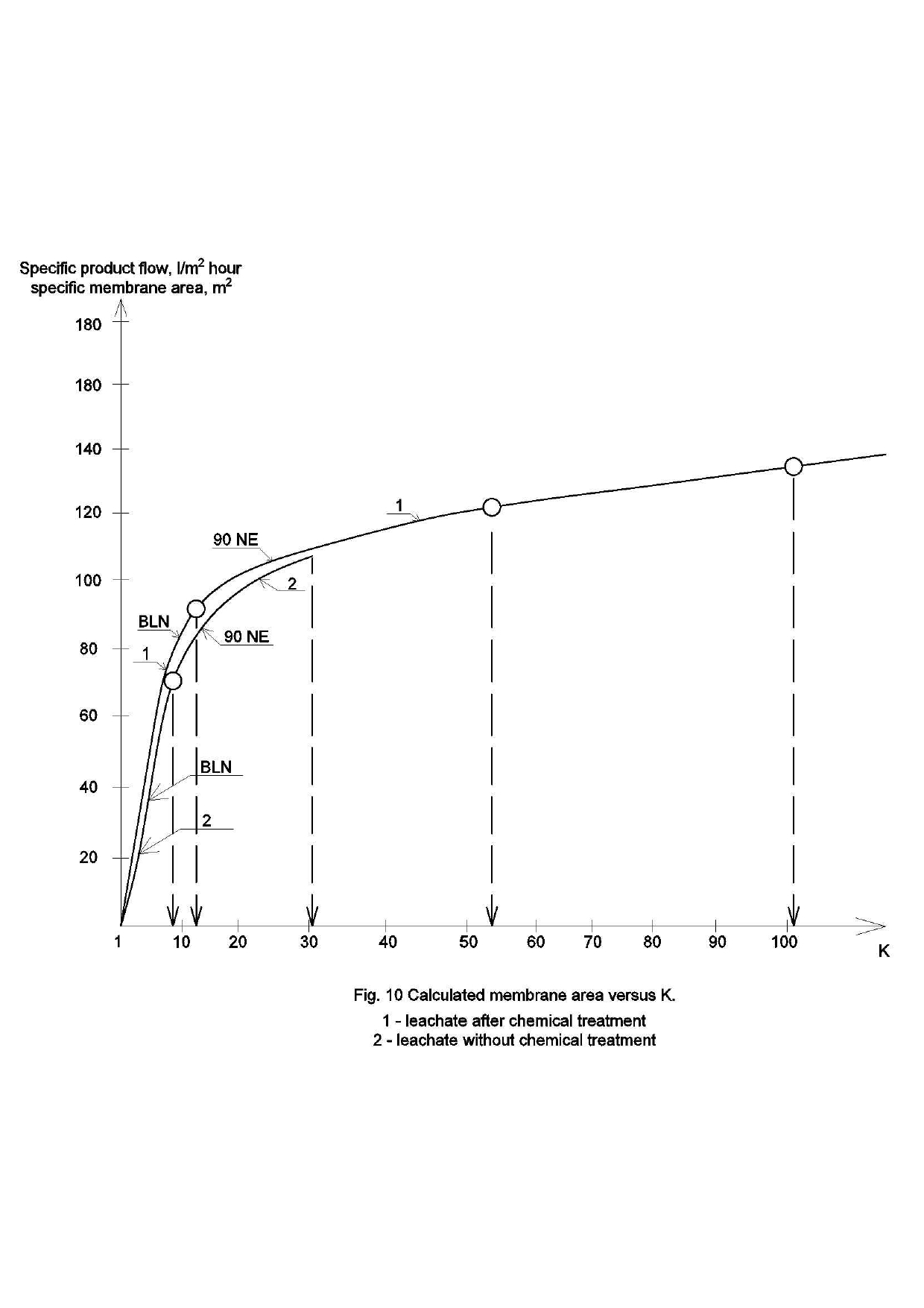
Main steps to calculate the required membrane area and amount of membrane modules during first stage of water treatment with BLN membrane when K is increased from 1 to 7 is illustrated on Figure 7. Figure 7 shows the amounts of product obtained in the K ranges from 1 to 3, from 3 to 4 and from 4 to 7. The obtained product amounts are presented as a percentage of the total volume produced during treatment of 70 liters of the feed wastewater with the final K value of 7 (after 60 liters of product water was produced). The selection of value 7 is explained by the low product water quality obtained with K values from 7 to 10. The calculations are made for the case when the required product flow of membrane facility is 1000 liter per hour and concentrate flow is 140 liters per hour. For each range of K value product flux was determined and the arithmetic average value of membrane specific flux was determined, expressed in: liters/hour / square meter (Figure 7).
Using calculated values of product water produced during one hour in the selected range of K and average specific value of membrane flux within this range, we can calculate the required membrane area to provide the required amount of product water during one hour (Figure 8). Calculations are made for the case when BLN membranes treat wastewater and K value changes from 1 to 7, and the pressure was 6 Bars. The required number of membrane elements can be determined assuming that membrane area in a standard 4040 element (BLN 4040 ) manufactured by CSM Company (Korea) equals 10 square meters. Thus, to decrease wastewater flow by 7 times and to produce 1000 liters of quality product water per hour) we need to use 12 elements of 4040 standard. The K value grows «step by step», therefore elements can be connected in series: 5 in parallel, then 4 and 3.
Both cases of treatment of leachate without chemical treatment and after chemical treatment are determined. For the case when leachate was not subjected to chemical treatment and is directly purified by reverse osmosis membranes, the first stage recovery value did not exceed 90 per cent due to the loss of product flow. Figure 8 demonstrates results of membrane area calculations as a dependence of specific required membrane area (to treat 1000 liters per hour of leachate) on K value reached during leachate treatment.
For the case when chemical pretreatment of leachate is not implemented, high recovery values are not reached due to high COD of leachate and low membrane flux (Figure 8). More 4040 BLN membrane elements are used to produce purified water and concentrate after BLN modules directly enters 90NE nanofiltration modules to further reduce concentrate flow to reach 90% recovery value.
To facilitate further increase of recovery during the third stage of nanofiltration increase, leachate first stage concentrate is collected in tank 11 where coagulation and flocculation of organics occurs. Coagulation and sedimentation of deposited organics required large volume of water treatment structures and footprint. For the case when leachate is «directly» treated by RO, the intermediate tank 11 and the third stage pump are not used. Analysis and studying of the experimentally obtained results allowed us to draw to the following conclusions.
- To reach the required ammonia concentration in product water, double stage treatment of feed with low pressure reverse osmosis membrane treatment is required. High recovery values are reached through the additional treatment of concentrate with nanofiltration membranes.
- Influence of dissolved organics defined as COD on membrane performance is investigated. Leachate COD decreases membrane flux and should be accounted when membrane area is determined. Flocculation and further sedimentation of organics prior to reverse osmosis treatment facilitates membrane filtration and provides higher values of recovery.
- Application of reverse osmosis and nanofiltration membranes combined with chemical leachate pretreatment enables us to produce quality water that meets discharge regulation standards and concentrate flow that does not exceed 0.3-0.5% of the total feed water that enters water treatment plant.
References
[1] Hussein L. Abdel-Shafy, Mona S.M.Mansour. Solid waste issue: Sources, composition, disposal, recycling and valorization. Egyptian Journal of Petroleum. Volume 27, Issue 4, December 2018, Pages 1275-1290
[2] J. Wiszniowski, D.Robert,J.Summer-Gorska, K. Miksh, J.V. Weber. Landfill leachate treatment methods:a review. Environ. Chem. Lett. (2006). 4:51-61. DOI 10.1007/s10311-005-0016-z .
[3] Sun W.Y., Kang M.S., Yim S.K. Choi K.H. Advances Landfill Leachate treatment using an integrated membrane processes. Desalination,(2002),v.149,p. 109-114.
[4] Amokrane A.,Comel C., Veron J. Landfill leachate pretreatment by coagulation -flocculation. Water Resourses.(1997), 31:2775-2782.
[5] Mark Wilf. The Guidebook to membrane for wastewater reclamation. Balaban Desalination Publications, 2010, V.788.
[6] A. Perez- Gonzales, A.M. Urtiaga, R, Ibanez, I Ortoz. State-of the art and review of the treatment technologies of water reverse osmosis concentrates. Water Research, 46 (2012)267-283.
[7] Pervov A.G., Andrianov A.P. Application of membranes to treat wastewater for its recycling and reuse: new considerations to reduce fouling and recovery up to 99 percent // Desalination and Water Treatment. 2011. Vol. 35 (1-3). Pp. 2-9. DOI: 10.5004/ dwt.2011.3133
[8] Pervov A. Application of reverse osmosis to treat and reuse petrochemical wastewater // The International Desalination Association World Congress — Sao Paolo, Brazil, REF: IDA17WC-57868 Pervov
[9] Alexei Pervov, Konstantin Tikhonov, Woiciech Dabrovski. Application of reverse osmosis to treat high ammonia concentrated reject water from sewage sludge digestion. Desalination and water treatment, www.deswater.com, doi: 10.5004 /dwt.2018.22009, (2018)1-9.
[10] A.G.Pervov, Xuan Quyet Nguen and E.B.Yurchevski. Investigation of the influence of organics contained in natural water on the performance of reverse osmosis membranes. ISSN 2517-7516, Membranes and membrane technologies, 2019, vol. 1, No 5, pp. 286-297. Pleiades Publishing, Ltd. 2019.